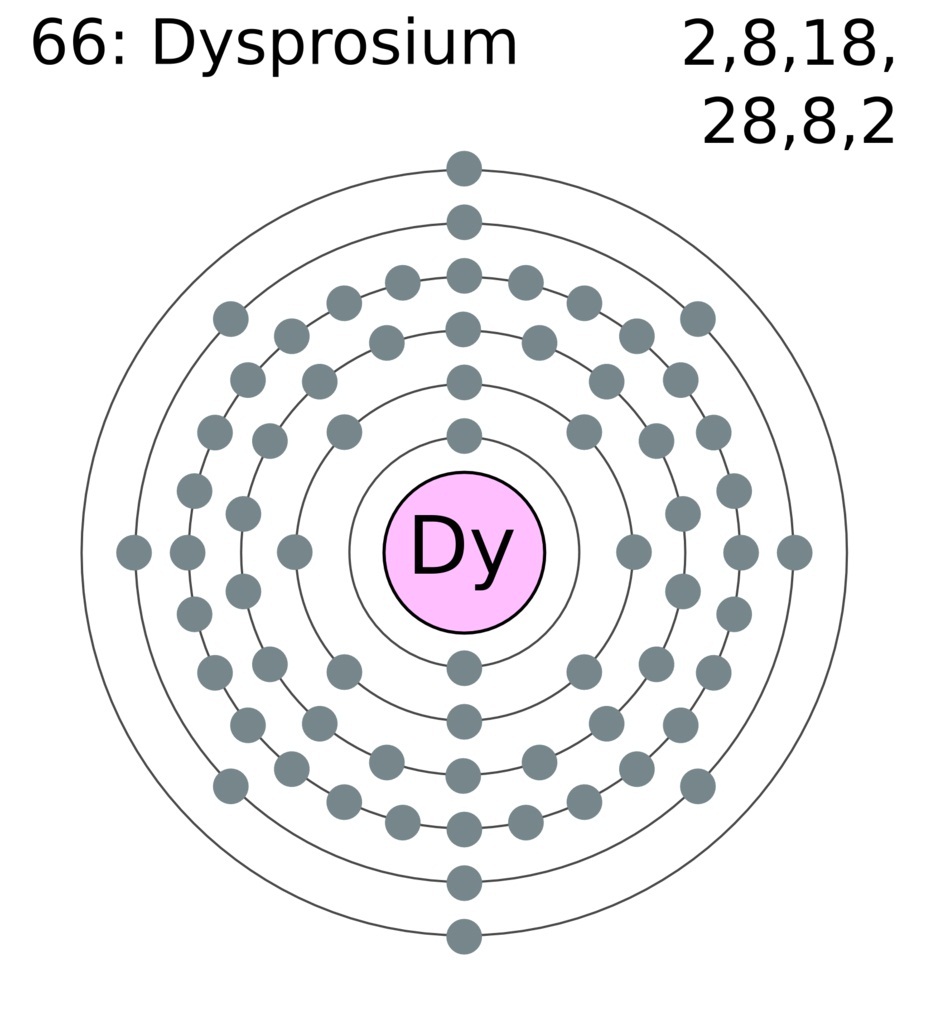
Update: Here’s a visualization of the categories above. For example, cadmium could not be C because that’s carbon, and it could not be Ca because that’s calcium. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images. Many of these elements would cause a conflict if they had been abbreviated using one of the above rules. Element Dysprosium (Dy), Group 19, Atomic Number 66, f-block, Mass 162.500. The next largest group of elements are abbreviated by their first letter and the next consonant, skipping over a vowel. Xenon could have been X, or dysprosium could have been just D, but that’s not how it was done. For example: actinium, aluminum, americium, and argon. 
There are several elements that start with the same letter, and no element uses just the first letter. For example, helium uses He because hydrogen already took H. Many of these elements use the first two letters to avoid a conflict with the first letter. When in doubt, guess the first two letters. The largest group of elements are those abbreviated by the first two letters of their name. The easiest abbreviations to remember are simply the first letters of the element names (in English). I included Tungsten in this section because it also has an abbreviation that is mnemonic in another language, in this case German. The elements that have been known the longest often have abbreviations that are mnemonic in Latin. Here’s a survey of how the elements are abbreviated. If you don’t know the abbreviation for an element, is there a simple algorithm that would let you narrow the range of possibilities or improve your odds at guessing? The electronic configuration of Dysprosium will be 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s2 5p6 4f10 6s2.I’ve wondered occasionally about the patterns in how chemical elements are abbreviated. It is however now an essential additive in NdFeB production. While it has one of the highest magnetic moments of any of the rare earths (10.6B), this has not resulted in an ability to perform on its own as a practical alternative to neodymium compositions. How do you write the electron configuration for Dysprosium? Dysprosium is most commonly used in neodymium-iron-boron high strength permanent magnets. The electronic configuration of Dysprosium will be 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s2 5p6 4f10 6s2. What is the electronic configuration of Dysprosium 66? What is the boiling Point of Dysprosium in Kelvin?īoiling Point of Dysprosium in Kelvin is 2840 K. Melting Point of Dysprosium in Kelvin is 1685 K. What is the melting Point of Dysprosium in Kelvin? What is the boiling Point of Dysprosium?īoiling Point of Dysprosium is 2840 K. Dysprosium has 66 electrons out of which 3 valence electrons are present in the 4f10 6s2 outer orbitals of atom. How many valence electrons does a Dysprosium atom have?ĭysprosium has 3 valence electrons. Dysprosium was first isolated by undefined in undefined. The element Dysprosium was discovered by P.E.L. What is the color of Dysprosium?ĭysprosium is of Silver color. It is located in group null and period 6 in the modern periodic table. Dysprosium is the 66 element on the periodic table. What is the position of Dysprosium in the Periodic Table?ĭysprosium is a chemical element with the symbol Dy and atomic number 66. Dysprosium is a chemical element with symbol Dy and atomic number 66. Click on the column header to sort the table by that column or click on an element name to get detailed facts about the element. To form abbreviated notation of electronic configuration, the completely filled subshells are replaced by the noble gas of the preceding period in square brackets. Here's a list of all of the chemical elements of the periodic table ordered by increasing atomic number. The abbreviated electronic configuration of Dysprosium is 4f10 6s2. What is the abbreviated electronic configuration of Dysprosium? The electronic configuration of Dysprosium is 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s2 5p6 4f10 6s2. What is the electronic configuration of Dysprosium? Optical Properties of Dysprosium Refractive IndexĪcoustic Properties of Dysprosium Speed of Soundĭysprosium Thermal Properties - Enthalpies and thermodynamics A Resource for Elementary, Middle School, and High School. Refer to table below for the Electrical properties ofDysprosium Electrical Conductivityĭysprosium Heat and Conduction Properties Thermal Conductivityĭysprosium Magnetic Properties Magnetic Type Los Alamos National Laboratorys Chemistry Division Presents. Hardness of Dysprosium - Tests to Measure of Hardness of Element Mohs Hardnessĭysprosium is Conductor of electricity. Refer to below table for Dysprosium Physical Properties DensityĨ.551 g/cm3(when liquid at m.p density is $8.37 g/cm3)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
